What Forms When Hydrochloric Acid and Potassium Sulfite React
How would potassium hydroxide react hydrochloric acid. Aqueous potassium phosphate was mixed with aqueous magnesium chloride and a crystallized magnesium phosphate product.

Potassium Sulphite On Reacting With Hydrochloric Acid Released Gas Cl2 So2 H2s
There are 2 atoms of Na on the left side of the equation and 1atom on the right side.

. When a solution of hydrobromic acid and potassium sulfite aq are combined potassium bromide water and sulfur dioxide gas are formed. K 2 S O 3 2 H C l 2 K C l H 2 O S O 2. Write a balanced chemical equation for this reaction.
Molecular equation and net ionic equation for the reaction of. Potassium sulphite on reacting with hydrochloric acid releases sulphur dioxide S O 2 gas. Write a balanced chemical equation to answer the following question.
Bones are mostly calcium phosphate. Hydrochloric Acid with CopperII Sulfate Your browser does not support the video tag. Suppose 4 moles of hydrochloric acid react with excess potassium carbonate.
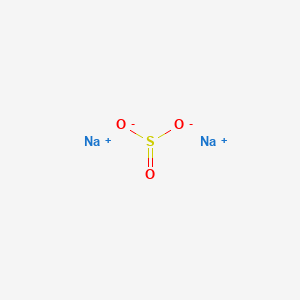
You can thus say that the balanced chemical equation that describes this. Dilute hydrochloric acid reacts with potassium sulfate so we can write the following equation for it. Aqueous hydrochloric acid reacts with aqueous sodium sulfite to form aqueous sulfurous acid and aqueous sodium chloride.
Sodium sulfite and hydrochloric acid. The reaction of an acid with a base is called neutralization. This is illustrated below.
What volume in L of gas is formed by completely reacting 445g of potassium sulfite at 134 atm and 221C. Aqueous sulfurous acid H2SO3 and aqueous sodium chloride are formed by the reaction of aqueous sodium sulfite Na2SO3 and aqueous hydrochloric acid HCl. A salt and water are formed.
Calcium chloride reacts with potassium phosphate to form calcium phosphate and potassium chloride. Suppose hydrochloric acid reacts with potassium sulfite yielding water sulfur dioxide and potassium chloride. The chemical equation for the reaction sodium sulfite plus hydrochloric acid isNa2SO3 2HCl -- SO2 H2O 2NaCl.
Acid base rarr Salt and water Explanation. 2HCl aq Na2SO3 aq 2NaCl aq H2SO3 aq Explanation. Sodium nitrate reacts with.
Solid potassium sulfite reacts with aqueous hydrochloric acid. Potassium hydroxide KOH reacts with hydrochloric acid HCl to produce potassium chloride KCl a salt and water H 2 O. Write the state s l g aq for each substance3.
See answer 1 Best Answer. During this reaction acid reacts with base to form salt and water. Chemistry questions and answers.
What forms when hydrochloric acid and potassium sulfite react. Here when calcium hydroxide reacts with hydrochloric acid it forms calcium chloride and water as products and if u want the ionic eq. This reaction is similar to the one of a carbonate and acid.
The reaction would yield potassium sulfite and hydrogen permanganateH2SO3. Similarly you may ask what type of reaction is potassium carbonate and hydrochloric acid. In this case sulphur-di-oxide gas S O 2 is evolved in the above reaction with the formation of potassium chloride.
Solid potassium sulfite reacts with aqueous hydrochloric acid. Write a balanced chemical. And here we got a strong base potassium hydroxide and a strong acid in aqueous solution.
Salt Reactions Whenever a strong acid reacts with a salt of a weak acid an exchange of ions occurs and the product comprises of the. Zinc sulfide and hydrochloric acid Molecular equation and net ionic equation for the reaction of. Sodium Hydrogen Sulfite reacts with hydrochloric acid to produce sulfur dioxide gas water and sodium chloride NaHSO3 HCl ----- SO2 H2O NaCl 2.
Les Ebdon EH. Calcium sulfite CaSO_3 will react with hydrochloric acid HCl to produce aqueous calcium chloride CaCl_2 and sulfurous acid H_2SO_3. The balanced equation for this reaction is CaCO3 2HCl CaCl2 H2O CO2.
HCl aq Na2SO3 aq NaCl aq H2SO3 aq Let us balance the equation. However sulfurous acid does not exist in aqueous solution as a molecule it actually decomposes to form sulfur dioxide SO_2 and water. How many grams of sulfur dioxide are produced.
CaOH2 2 HCl ----- 2 CaCl2 2 H20. Here copperII sulfate CuSO 4 is added to hydrochloric acid HClThe. 1 Answer anor277 Apr 21 2018 This one is easy.

How To Balance K2so3 Hcl Kcl So2 H2o Potassium Sulfite Hydrochloric Acid Youtube

Potassium Sulphite On Reacting With Hydrochloric Acid Released Gas Cl2 So2 H2s

Solved 4 Test For The Sulfite Ion 5032 The Sulfite Chegg Com

State Your Observations When Dilute Hydrochloric Acid Reacts With Sodium Sulphite

Solved Complete The Following Questions 1 Sodium Hydrogen Chegg Com
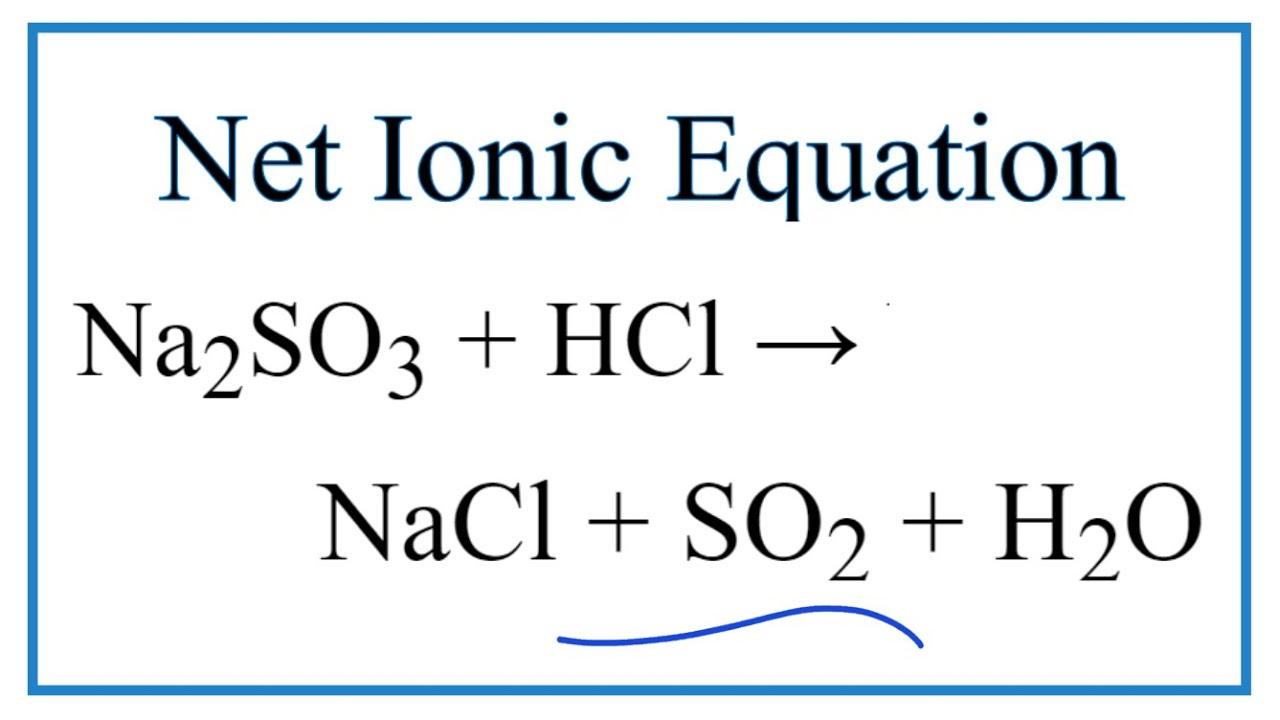
How To Write The Net Ionic Equation For Na2so3 Hcl Nacl So2 H2o Youtube
/5158DFA1913FDEFA802586B600762FEA/$file/KAA10215_structure.png)
Sodium Sulfite Heptahydrate 10102 15 5 Biosynth Carbosynth
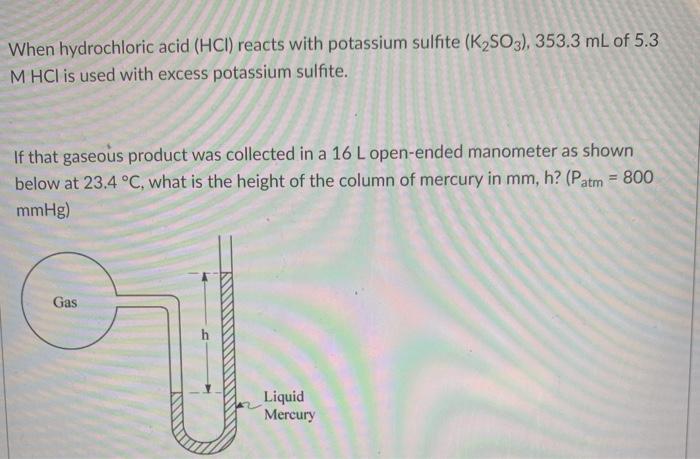
Solved When Hydrochloric Acid Hci Reacts With Potassium Chegg Com
/CE0E94FB611B330D802585F90081BB81/$file/FS46366_structure.png)
Sodium Sulfite Anhydrous Ph Eur 7757 83 7 Biosynth Carbosynth
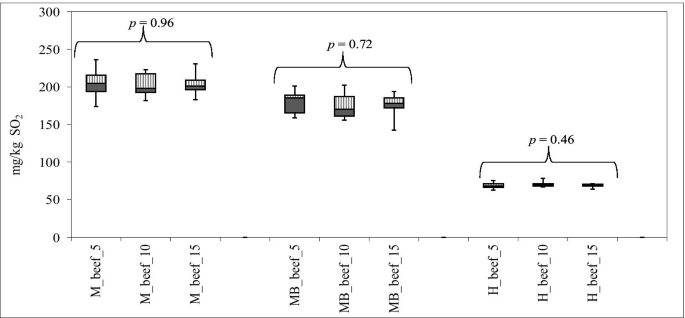
Sulfites In Fresh Meat And Meat Preparations Commercialized In Portugal Springerlink
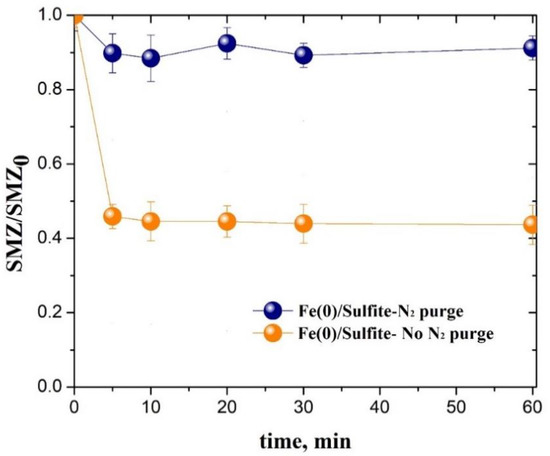
Water Free Full Text Comparative Study Of The Oxidative Degradation Of Different 4 Aminobenzene Sulfonamides In Aqueous Solution By Sulfite Activation In The Presence Of Fe 0 Fe Ii Fe Iii Or Fe Vi Html

How To Balance K2so3 Hcl Kcl So2 H2o Potassium Sulfite Hydrochloric Acid Youtube

How To Balance K2so3 Hcl Kcl So2 H2o Potassium Sulfite Hydrochloric Acid Youtube

How To Balance Na2so3 Hcl Nacl So2 H2o Sodium Sulfite Hydrochloric Acid Youtube

Establishing A Method Of Hplc Involving Precolumn Derivatization By 2 2 Dithiobis 5 Nitropyridine To Determine The Sulfites In Shrimps In Comparison With Ion Chromatography Yang 2019 Food Science Amp Nutrition Wiley Online Library

Question Video Identifying An Unknown Solution From The Reaction With A Sodium Sulfite Solution Nagwa

E226 Calcium Sulphite Food Additives Nutrient Calcium

Solved When An Aqueous Solution Of Hydrochloric Acid Chegg Com
Comments
Post a Comment